After taking a long break from blogging but definitely not reading about science, I have decided to start off the academic year by attending the Royal Society of Medicine’s second innovation summit this year to get my mind in the right headspace.
As always, listening to the speakers truly inspires me and opens my eyes to the vast possibilities of Medicine. Throughout the day I heard from those discussing tackling global health challenges, technology in the applied medical setting and innovative research and I will share with you four of the talks I heard about.
1.
Blood bank shortages is an ongoing problem with around 40% fewer blood donation volunteers than a decade ago in the UK. A new ‘natural’ lab grown source of blood being developed by Dr Joanne Mountford and Professor Marc Turner who were granted the £5 million Strategic award by the Scottish National Blood Transfusion Service could potentially solve this issue.
Current obstacles that need to be overcome with donated blood are; immune incompatibility, sufficiency, transfusion transmitted infection and iron loading (seen in those with β thalassemia form multiple blood transfusions). The researchers use donated skin cells (fibroblasts) which are then genetically reprogrammed forming induced pluripotent cells which are able to self renew and expand indefinitely. By using iPS cells rather than embryonic stem cells collected from unused IVF embryos, it avoids the ethical considerations to think about and the possibility of gene expression. It was also important to them to make sure the blood was ‘natural’ and not made form synthetic materials.
In the lab they are currently able to grow blood with fetal haemoglobin expression (α/γ chain) which is viable in adults and due to their young nature they could give a longer therapeutic benefit and reduce the frequency of transfusions whereas blood bags contain a mixture of cell ages. The cells are matured in suspense cell cultures using growth factors and cytokines with the entire process taking about 30 days.
The aim is to create O Rhd negative blood which can be given to 98% of the population, however challenges they spoke about included overcoming membrane instability after enucleation. It is essential that the nucleated normoblast is converted to a biconcave shape giving them a larger surface area to absorb oxygen. This is checked by marking the proteins with fluoresce to see via laser or stain under a microscope. Furthermore, at the moment cells are only grown to obtain protein and not for the actual cell component so volume reduction and separation technology needs to be developed in order to scale up the RBC production. To meet clinical requirements, 2×1012 RBC would need to be generated, found in a standard red blood cell concentrate.
They have come a long way since starting this research as they are now able to make 5ml of blood (10% of a unit) efficiently and hope to start the first human trial in late 2016. This concept of natural (non-synthetic) blood looks to be an innovative tool in the future with the ability to reduce NHS costs in disposing of unused donated blood and delivering to developing countries where blood loss during childbirth can save hundreds of thousands of lives.
2.
French company DBV Technologies are revolutionising food allergy treatments by developing the Viaskin patch. Current immunotherapy treatment for food allergies (to build up tolerance) are by subcutaneous injections which can lead to itchy and swollen skin. This patch administers the allergen to intact skin avoiding contact with blood increasing tolerability therefore lowering the risk of a systemic, allergic reaction in case of chance exposure.
Viaskin is a non-invasive electrostatic patch that hopes to be a technique to desensitise and improve patient’s tolerability by making the immune system used to the allergen by placing small amounts of the allergen onto intact skin.
The patch consists of a dry allergen layer, a condensation chamber to increase permeability of the skin and solubilises the antigen allowing it to penetrate through the epidermis. Here the antigen is able to reach Langerhan cells which leads to draining of lymph nodes, antigen processing and presentation to T cells which stimulate the immune response and T memory cells. This method uses a passive pathway with no contact with blood to avoid an allergic reaction.
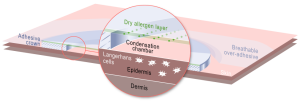
Preliminary tests done by DBV show that epicutaneous immunotherapy (EPIT) may induce an epigenetic change (specifically a modification of DNA methylation).
Dr. Pierre-Henri Benhamou, Chairman and CEO of DBV Technologies has previously said that, “EPIT appears to be the only method of specific immunotherapy able to confer durable protection in case of accidental exposure to allergens. We believe that this is probably due to the ability to influence the immune response to allergens at the genomic level”.
A 2014 study published in the British Society for Allergy and Clinical Immunuology concluded that regulatory T cells (Tregs) mediated mechanism of EPIT resulted in long-term protection against eosinophilic disorders* in peanut-sensitised mice which is a very positive outcome.
As it is easy to use and safe, early intervention in children could be a realistic possibility without the risk of anaphylaxis could prevent the disease developing. With over 5 million peanut allergy sufferers in Europe and the USA, if successful could make a huge difference to their lives. DBV Technologies are also currently working on using Viaskin technology to treat milk and egg allergies and an alternative to some vaccines.
*Too many eosinophils (WBCs) produced causing chronic inflammation and tissue damage.
3.
All the way from Michigan, Glenn Green and Scott Hollister came to speak about just the second procedure of it’s kind to use a custom biodegradable implant. The 3 year old patient’s condition (18 months old at the time) had left him hospital bed-ridden with ventilator levels at their maximum pressure and was even placed in a medically-induced coma to keep him alive. The condition the child had is called, Tetralogy of Fallot with absent pulmonary valve syndrome, only 3% of those with Tetralogy of Fallot also missing the pulmonary valve.
Due to the absence of the pulmonary valve, pulmonary regurgitation occurs (retrograde flow of blood from pulmonary artery to the right ventricle). This lead to him developing severe tracheobronchomalacia, a disease that weakens the muscles around the trachea. It was so debilitating that he would turn blue sometimes four to five times a day as the smallest movement could cause his airways to collapse to the point where they were just small slits. There are approximately 1 in 2,200 babies born with tracheobronchomalacia but severe cases are rare.
In December 2013, his condition was worsening and plans needed to be finalised with emergency clearance by the FDA to create and implant a bioresorbable tracheal splint made from a biopolymer. After taking a CT scan of the child’s trachea and bronchi, the splint was produced using a system that takes an image computer model alongside 3D laser printing to create a custom-design. They had about 20 to 30 different models as they knew how it looked on screen but were prepared for any contingencies.
In 2014 Green assisted Dr. Richard Ohye in performing the surgery, two splints were sown around his left and right bronchi to allow expansion of the airways and support to aid growth. Since the operation, the boys airways have remained open and ventilation levels are less than a quarter of what they used to be. It shall take around three years for the trachea to remodel itself and that is about how long it will take for the splint to be reabsorbed.
4.
In May 2015, surgical teams from Houston Methodist hospital and the University of Texas MD Anderson Cancer centre collaborated to perform the first composite vascularised allo-transportation surgery alongside craniofacial tissue transplant.
A 55 year old man presented with a failing kidney and pancreas from a previous transplant in 1992 as a result of Type 1 diabetes and a previous history of leiomyosarcoma (rare cancer of soft muscle tissue) which left him with a large, deep wound on his scalp and exposure of the skull bone. Due to the risk of infection from his head wound, surgeons were hesitant to perform a new organ transplant, however Dr. Jesse Selber saw an opportunity here. As the patient was already immunosuppressed from previous solid organ transplants, the high threshold that ordinary transplant candidates would have to meet due to increased risk of taking long-term immunosuppressants would not be a problem. Carrying out a skull and scalp transplant simultaneous to the double-organ transplant would mean the anti-rejection medication could support them all.
A wait of 18 months ensued to find the correct donor as he already had other antibodies in his blood from the previous transplant and many donors could not be used. Selber’s skills in microvascular technique were essential to this type of surgery as blood vessels 1/16th of an inch need to be connected to the scalp quickly to restore circulation. Transplants involving tissue structures such as skin, bone, blood vessels and nerves such as this one are called vascularised composite allograft and techniques which are still relatively new and techniques are still progressing. Dr Selber expressed to us that while this surgery took many months of meticulous planning and organising, they had to improvise after realising that the donor’s skull was much thicker compared to the remainder of the patient’s and had to reshape the skull with saws and drills in order to make it fit. Being the first surgery of it’s kind, the surgeons were amazed when seeing the blood flow through the scalp once it had been connected as the grey organ was brought to life. The pancreas and kidneys were then transplanted with surgeons having to work around the calcifications formed from his previous failing organs. After 15 hours, the four transplants were complete and the patient continues to be closely monitored using Skype consultations as a method of keeping up to date so no travel time is needed unless in emergency.
Dr. Jesse Selber also spoke about how he has now asked the patient to take daily photographs of his head to oversee the skull and scalp transplant healing to get a good overview. Selber recalls his fascination at watching the patient’s scalp sweat the Wednesday after the surgery in a hot recovery room and how it was unexpected due to the nature of not knowing what is to come as it is a new type of surgery. The doctors expect the patient to have rejection episodes, as is common with allografts. In fact just a few weeks ago there was a scare that the body might be rejecting the scalp as a large rash was appearing on his scalp however after testing and a revising of medication there are no signs of rejection. This surgery was innovative and very complex with around 50 health professionals all gathered in one surgical theatre to make this possible.
Hope this has given you a glimpse and want to go out and innovate!
Am aflat aceasta pagina, dupa ce am cautat despre RSM 11th Medical Innovation summit pe Google.
Se pare ca informatia dvs e foarte valoroasa, mai ales
ca am mai gasit aici si despre ora, ora exacta, lucruri interesante si folositoare.
Mult succes in continuare!